Today was a slightly less busy Monday than usual since I was at lab for a gazillion hours this weekend (ok slight exaggeration but longer than usual, and got a bit ahead of usual). This meant not only I had time to mull some things over while doing cell culture less hurriedly than usual on a Monday, but I had a little half hour or so before lunch to look at my weekly pubmed email updates.
A new paper in the Annals of Oncology caught my attention since it was a large population on IBC patient outcomes study regarding ~7700 patients diagnosed between 1990 and 2010. This caught my attention because there hasn’t been much written on this lately outside of single institution studies (such as MDACC’s own experience) – which have limitations including positive bias (places with terrible outcomes less likely to write academic papers vs centers of excellence with nuggets to share). Intrigued I read over most of it, and scrutinized the figures/tables. The purpose of the paper was to investigate whether overall outcomes of IBC have improved over time using a national healthcare database called SEER, focusing on stage 3 patients. Just to share a piece of the promising data they show, figure 1 shows a nice positive trend in 2-year overall survival that goes from 50% in 1990 to ~70% in 2010. However as I dug deeper, a few things disturbed me. The data in most of the tables is stratified by year of diagnosis…..a few things that stood out to me.
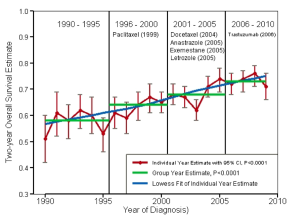
Figure 1: 2 year overall survival and the years in which selected drugs became used in IBC.
Ok first I have to explain a few things– today’s definitive treatment for stage 3 IBC given with curative intent includes upfront (neoadjuvant) chemotherapy, then a modified radical mastectomy followed by radiation. If HER2 positive, 1 year of Herceptin is included starting concurrently with chemotherapy (finished after surgery). If ER+, 5+ years of adjuvant endocrine therapy appropriate to menopausal status (tamoxifen or aromatase inhibitor) is given after radiation.
Going back to the SEER data – what do these data show? In the most recent 5 year period specifically, only 82% of patients received a mastectomy. 18% rate of breast conserving surgery is unacceptable when IBC is an absolute contraindication to lumpectomy (the reason being since the disease by definition involves the skin, it must go as well as the whole breast!). These surgeries also were not done to today’s standard. In the table there is a section on number of nodes examined. Shockingly, 20.5% had ZERO nodes examined. Mind boggled! Axillary nodal dissection is an important component of comprehensive staging and is considered a very standard step during surgery. While sentinel node dissection (SLND) is becoming more and more considered in other locally advanced breast cancers treated by mastectomy, the role of SLND is still under investigation in IBC. The MDACC surgical team is trying to identify a subset of patients who can be spared the morbidity from axillary dissections yet adequately stage their axilla. However as of today, every patient should have 1 or more nodes examined. Preferably all the level 1 and 2 nodes that the surgeon finds.
Next up – only 51% of patients received radiation. This should be 100%. No excuses – radiation is a very important form of local therapy that has been proven to make a difference in outcomes. Studies of many high-risk breast cancer patients have demonstrated that radiation decreases the risk of loco-regional recurrence by 50-66% – which is a lot when your baseline recurrence risk is as high as it is for most IBC patients.
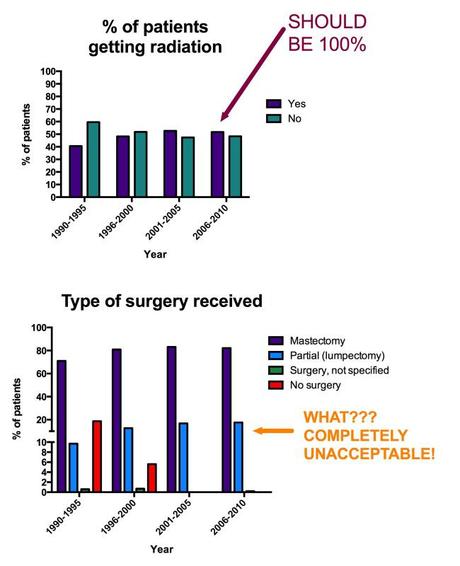
Figure 2: Data showing less than optimal treatment provided.
Lastly, but not of least importance, is an analysis of outcomes by race. For white patients diagnosed between 2006 and 2010 – the 2-year OS is 75%, whereas among black women it is only 61%. Why is this? Complicated topic – likely both underlying biology as well as factors relating to access to good quality care. Another topic for another day, but just another statistic from the paper that bothered me, and barely commented on by the authors.
So back to the original question are we doing better? Yes statistically the p-values for all the comparison show an increase in outcomes over time. But there is still a lot to do. Clearly triple-negative IBC still lags behind – less than 15% have a pathological complete response and this leads to a median overall survival around 2 years. To do better in TN-IBC, we will not only need to increase pCR rates (perhaps by adding targeted agents to chemotherapy or changing cocktails to take advantage of new knowledge) but also find some adjuvant therapy that is useful to mimic the benefits from endocrine therapy in ER+ disease. What about a vaccine or similar immunotherapy approach as well.
Perhaps it would be best to start with applying what we already know though. Outcomes are best with a multi-disciplinary team working together – good upfront staging (radiologists working hand-in-hand with pathologist to get enough of the correct tissue), optimal chemotherapy (anthracycline+taxane or otherwise) upfront with a competent medical oncologist closely following response. The medical oncologist then handing the patient off to trusted surgical oncologists who will do the CORRECT surgery, get clean margins and then of course a radiation oncologist to mop up any residual local disease with an appropriate dose, frequency and fields based on the individual patients risk factors. It really shouldn’t be up to a patient newly diagnosed with an uncommon, rapidly progressing life-threatening disease to know this stuff. Are you with me?
Source: Dawood S et al, “Survival of women with inflammatory breast cancer: A large population based study”. Annals of Oncology 2014 (in press), article link
