There is good news for women with dense breasts. The Food and Drug Administration (FDA) has approved the first ultrasound device to detect breast cancer in women with dense breast tissue.
The FDA announced, this past Tuesday, that this new device is to be used along with standard mammography for women with no symptoms of breast cancer.
The U.S. National Cancer Institute puts the number of women with dense breasts at 40%. Dense breasts are problematic because they can make standard mammograms more difficult to read. Dense breasts have more connective and glandular tissue than less-dense breasts, which contain more fatty tissue.
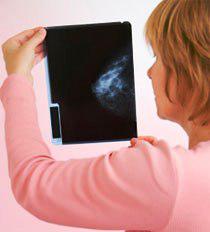 In a news release, the FDA stated that the problem with dense breasts is how they appear on regular mammograms. Dense breast tissue appears as solid white areas and so do tumors. Smaller tumors can be missed in dense breast tissue. According to the FDA, using high-frequency sound waves and computer analysis, however, ultrasound can detect tumors even in dense breasts.
In a news release, the FDA stated that the problem with dense breasts is how they appear on regular mammograms. Dense breast tissue appears as solid white areas and so do tumors. Smaller tumors can be missed in dense breast tissue. According to the FDA, using high-frequency sound waves and computer analysis, however, ultrasound can detect tumors even in dense breasts.

The somo-v Automated Breast Ultrasound System (ABUS), with a specially shaped transducer, can scan an entire breast and produce several images for physician review in about a minute
Approval for this new system came following a study of 200 healthy women with dense breasts and negative mammograms (ones that did not show cancer). Board-certified radiologists reviewed mammograms alone or along with ABUS images. Significantly more breast cancers were detected when ABUS and mammograms were both used.

