 Barbara Low in California, 1947. Credit: Low estate.
Barbara Low in California, 1947. Credit: Low estate.Barbara Low, a former research fellow for Linus Pauling and an esteemed scientist, died earlier this year at the age of 98. Low spent most of her career as a researcher and professor at Columbia University’s Vageos College of Physicians and Surgeons. She is perhaps best known for her work with protein structures, particularly her work on the structure of penicillin and her discovery of the pi-helix.
Barbara Wharton Rogers was born in Lancaster, England on March 23, 1920 (she married in 1950 and changed her name thereafter). After receiving her B.A. from Somerville College – an Oxford women’s college – in 1942, she went on to earn an M.A. and D. Phil. from Oxford University. As a component of her education, Low learned the techniques of x-ray crystallography, a field within the chemical sciences that was emerging for women. A major reason for this trend was the fact that one of the leading crystallographers of the era, Oxford professor Dorothy Crowfoot Hodgkin, was banned from teaching to men, so instead she taught crystallography to women at Somerville.
Low was one of Hodgkin’s star pupils, and after Low received her B.A. in chemistry, Hodgkin became Low’s advisor for her graduate studies. It was during these years that Hodgkin and Low determined the structure of penicillin using x-ray crystallography. In 1964, Hodgkin was awarded the Nobel Prize in chemistry for her work determining the structures of various important biochemical substances, penicillin certainly among them.

While she was working on her doctorate, Low spent a year at the California Institute of Technology as a research fellow, supervised by Linus Pauling. This was the start of what would become a fruitful and mutual working relationship between Pauling and Low. After leaving Caltech and graduating from Oxford, Low took a position as a research associate, and later as assistant professor of physical chemistry, at Harvard University. As her career advanced, Low kept in touch with Pauling and this connection proved beneficial on more than one occasion.
In the early 1950s, Low began to apply x-ray crystallographic techniques to a study of the structure of insulin. She did so during a period of much debate within the scientific community about the structure of various proteins. Pauling famously solved a piece of this puzzle in April 1951 when he published, “The structure of proteins: Two hydrogen-bonded helical configurations of the polypeptide chain” with collaborators Robert Corey and Herman Branson. In this paper, Pauling described for the first time the alpha helical structure of many proteins, a watershed moment that ushered in a whole new era of understanding across the discipline.
Barbara Low was, of course, also working on the structure of proteins, and she became particularly inspired to investigate the connection between structure and function after attending a lecture that Pauling gave at the Massachusetts Institute of Technology in March 1951. Low believed, as did Pauling, that the configuration of the folding of the protein was of more importance to its function than was the molecular make-up itself. Determined to apply this belief to her work on the structure of insulin, Low wrote several letters to Pauling asking him to verify the bond angle distances for the proteins about which he had lectured. Pauling gladly supplied Low with the requested data, even noting that he had double-checked the calculations as he was writing her back. Pauling also helped Low to secure scientific models for the structures that he had described.
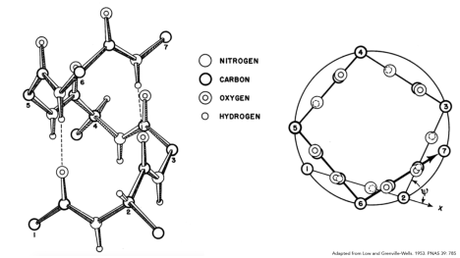
These data and models proved vital to one of Low’s most famous discoveries: the pi-helix. Like the alpha-helix, the pi-helix is a type of structure found in some proteins, though one that was not published by Pauling as part of his alpha-helix investigations. This failure may have been due to the pi-helix’ small size, which at the time of its discovery led some researchers to believe it to be an infrequent and rare structure. More modern day findings indicate however that the pi-helix is much more common than previously thought; present in about 15% of protein structures all told.
Low wrote about her discovery to Pauling shortly after the news was made public and received a mixed reply from her former mentor. At the beginning of his response, Pauling suggested that the pi-helix was most likely something that he “too ran across a while back” but acknowledged that Low’s structure was not “intermediate between the alpha helix and the gamma helix,” and thus both novel and genuine. The letter concludes with an admission from Pauling that his researchers may have “overlooked it” in their previous work.
Pauling’s hedging congratulations in this instance did not seem to negatively impact the duo’s relationship, and throughout their correspondence one intuits that the colleagues remained on friendly terms throughout the years. In many letters to Pauling, Low often concluded by giving her regards to Ava Helen. Low also developed a love for the comic Li’l Abner by way of Pauling, who had introduced her to the satirical strip at a dinner party in the early 1950s.
Pauling and Low were also, at times, involved in one another’s careers. When Pauling was denied a passport to travel to the Royal Society Meeting to attend the Protein Symposium in 1952, Low wrote to express her “shock” and to express how “shaken” she was that he had been treated this way. For his part, Pauling helped Low to secure grants and funding through multiple letters of support.
Pauling also provided assistance to Low as her research position at Harvard came to an end in June 1956 by putting her in contact with colleagues Detlev Bronk of Johns Hopkins University, John Kirkwood of Yale University, and DeWitt Stetten at the National Institute of Arthritis. While it is unclear how influential these contacts may have been in Low’s gaining her eventual position at Columbia, it is certainly worth noting that Stetten had recently left Columbia after having served there for nine years as an instructor of biochemistry.
However it came to pass, Low started at Columbia in 1956 as an assistant professor and was promoted to professor in 1966. She formally retired from Columbia in 1990, but stayed on as a lecturer until 2013. Like Pauling, Low was active both socially and politically, devoting significant time and energy to affirmative action activities at her institution. She passed away on January 10, 2019 at her home in the Bronx, New York.
