
Linus Pauling showing a molecular model to a young boy. 1950s.
[Part 3 of 3]
On March 31, 1951, Linus Pauling and numerous associates published seven revolutionary papers in a single issue of the Proceedings of the National Academy of Science. The research had been funded by the Rockefeller Foundation and carried out at the Gates and Crellin Laboratories of Chemistry, at Caltech. The first two articles: “The structure of proteins: Two hydrogen-bonded helical configurations of the polypeptide chain” and “Atomic coordinates and structure factors for two helical configurations of polypeptide chains,” have been discussed by us in the two weeks prior to this one. The remaining five will be described here in much shorter detail, as they are technical in the extreme.
The third article was titled “The structure of synthetic polypeptides,” and was written by Pauling and Robert B. Corey. The article claimed that the gamma helix and alpha-helix protein structures had forms that were also assumed by synthetic polypeptides. The authors discussed how the fibers of synthetic polypeptides had been analyzed using x-ray and infrared spectroscopy, which allowed them to determine the shape of the synthetic structures. Other scientists had also proposed the shapes of such structures, but Pauling and Corey rejected their hypotheses, as the structures the other scientists had proposed would have been “inherently unstable.” They concluded that their structure was the superior idea, and that while other structures potentially existed, they would be extremely difficult to measure due to their size.
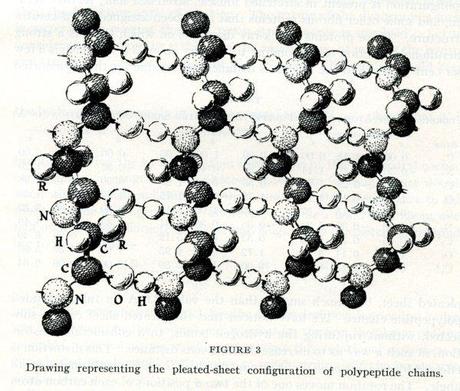
The fourth article was more crucial to the narrative of protein structure that Pauling and his collaborators were weaving. The title was self-explanatory, and somewhat less technical than the others: “The pleated sheet, a new layer configuration of polypeptide chains.” In it, Pauling discussed how it had been long-believed that polypeptide chains are fully stretched and bound to adjacent, lateral chains of protein. He proposed instead a new idea, the so-called “pleated sheet.” In his suggested structure, the chains formed planes and certain bonds were arranged perpendicular to the planes of the chain, instead of coincidental with them. As a result, the chains are staggered and scrunched, instead of stretched in long, parallel lines. The rest of the article was devoted to the mathematics that Pauling had used to develop and explain the shape.
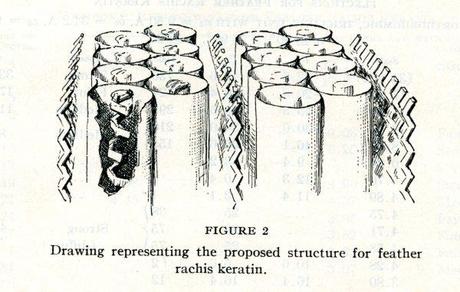
Feathers – specifically the atomic structure of feathers – was the topic of the fifth article, titled “The structure of feather rachis keratin.” The piece was written once again by Pauling and Corey and it analyzed rachis – a term with many meanings, but in this context referring to the central shaft of a feather – and keratins, which are structural proteins. The authors wrote that x-ray analysis of feather rachis keratin had shown the patterns of the polypeptide chains to be extremely complex, and notably shorter than expected. The rest of the article was spent explaining how the concept of the pleated sheet was mathematically relevant to feather rachis keratin.
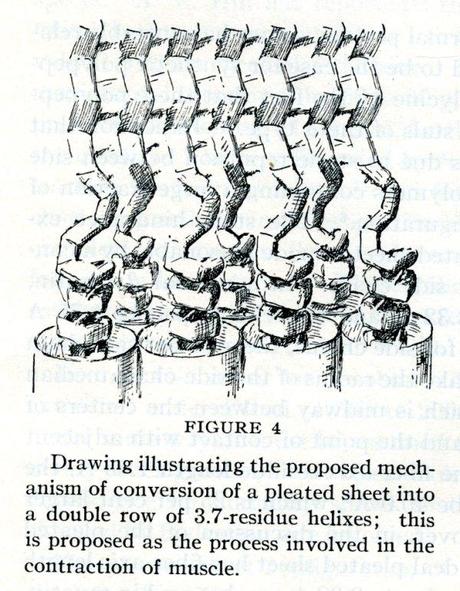
Second to last was an article called “The structure of hair, muscle, and related proteins,” written by Pauling and Corey. In it, the authors pointed out that it had been many years since R.O. Herzog and Willie Jancke, in 1926, had made important x-ray photos of hair, muscle, nerve and sinew. Pauling and Corey felt that these photos, though revolutionary, were no longer adequate. Yet despite this deficiency, few modern attempts had been made to take better photographs. Two scientists named Lotmar and Picken had tried in 1942, but Pauling felt that their pictures were likewise not detailed enough. The Caltech researchers determined that their lab had found enough data though, and proposed structures for hair, muscle and “related proteins.”
This article differed from the other six in that it had an addition dated April 10, 1951. Written by Verner Schomaker, the addition revealed that subsequent research had shown that, while its basic premise was correct, the argument outlined on the piece’s first two pages was in fact wrong, and that the rest of the article hoped to amend that. Pauling and Corey argued that relaxed muscle was configured as a sheet, while contracted muscle formed an alpha-helix. The sheet configuration was inherently unstable relative to the alpha-helix, which made it easy for the hydrogen bonds holding the muscle in a sheet to break. This breakage allowed the polypeptide chains to coil and in turn made the muscle contract. The mechanism to prevent a chain reaction that might result in the sheet ripping itself apart during contraction was not understood, though Pauling had some ideas for that as well. The rest of the article was spent analyzing the amounts of energy released in frog muscle contractions to provide hypothetical amounts of energy expenditure and size for contractions in human muscle.
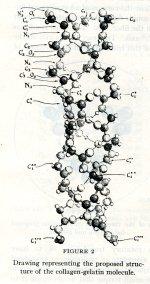
Representation of the collagen-gelatin molecule. April – May 1951.
The final article was “The structure of fibrous proteins of the collagen-gelatin group.” In it, Pauling wrote of his particular fascination with the protein in question:
Collagen is a very interesting protein. It has well-defined mechanical properties (great strength, reversible extensibility through only a small range) that make it suited to the special purposes to which it is put in the animal body, as in tendon, bone, tusk, skin, the cornea of the eye, intestinal tissue, and probably rather extensively in reticular structures of cells.
Another intriguing feature of collagen-gelatin was that it provided similar x-ray photos regardless of the source. In his article, Pauling noted that twenty-six samples, ranging from demineralized mammoth tusk to sheep gut lining, were all photographed by a scientist named Richard Bear and each resulted in remarkably similar images. Pauling compared them to a photograph of raw kangaroo tendon taken by Corey and Ralph W. G. Wyckoff, which also provided a view of what appeared to be the same structure. Pauling wrapped up the article discussing how three molecular chains wrapped into a distorted coil, and how the correlations between collagen-gelatin proteins and hydrogen could affect the structure.
The proteins work published by Linus Pauling and his Caltech colleagues in 1951 shook the scientific community and only added to Pauling’s growing fame. However, as time passed, evidence began to mount that his proposals regarding the gamma helix, muscle, and feather rachis were, in fact, wrong. Additionally, J.D. Bernal‘s lab found that the alpha helix, while fitting Pauling’s structural model, actually played a much smaller role in globular proteins than Pauling had suggested. However, Pauling’s media savvy and undeniable charisma won the day, at least in the short term. And so it was that, in the fall of 1951, (quoting Thomas Hager)
the 5 million readers of Life opened their new issues to find an enormous photo of Pauling, a big grin on his face, pointing to his space-filling model of the alpha helix. The headline read, ‘Chemists Solve a Great Mystery.’
